Click on images to enlarge
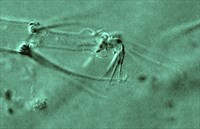
Fig. 1. Tetranychus urticae adult female (non-type) - detail of empodium I.
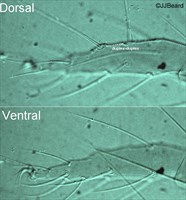
Fig. 2. Tetranychus urticae adult female (non-type) - detail of tarsus I indicating four proximal setae.

Fig. 3. Tetranychus urticae adult female (non-type) - detail of tarsus I indicating four proximal tactile setae.
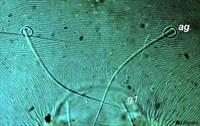
Fig. 4. Tetranychus urticae adult female (non-type) - detail of pattern of pregenital striae, indicating longitudinal striae.

Fig. 5. Tetranychus urticae adult female (non-type) - detail of pattern of pregenital striae, indicating longitudinal striae with weak to broken striae mesally.

Fig. 6. Tetranychus urticae adult female (non-type) - detail of pattern of pregenital striae, indicating broad anterior band of broken striae.
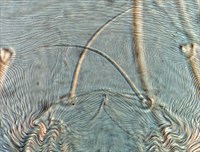
Fig. 7. Tetranychus urticae adult female (non-type) - detail of pattern of pregenital striae, indicating broad anterior band of broken striae.
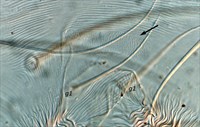
Fig. 8. Tetranychus urticae adult female (non-type) - detail of pattern of pregenital striae, indicating broad anterior band of broken striae.
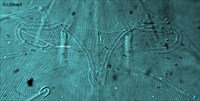
Fig. 9. Tetranychus urticae adult female (non-type) - detail of peritreme.
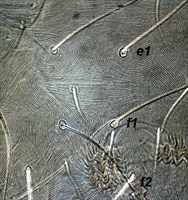
Fig. 10. Tetranychus urticae adult female (non-type) - detail of pattern of dorsal striae between setae e1 and f1.
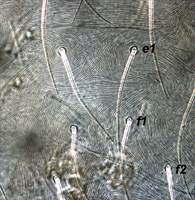
Fig. 11. Tetranychus urticae adult female (non-type) - detail of pattern of dorsal striae between setae e1 and f1.
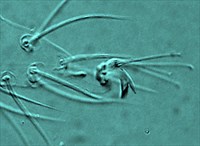
Fig. 12. Tetranychus urticae adult male (non-type) - detail of empodium I.
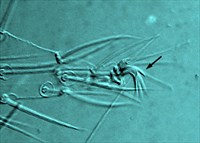
Fig. 13. Tetranychus urticae adult male (non-type) - detail of empodium II.
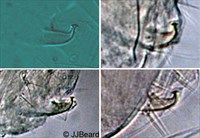
Fig. 14. Tetranychus urticae adult male (non-type) - detail of aedeagus.

Fig. 15. Comparison of the aedeagi of Tetranychus urticae, T. parakanzawai, T. pueraricola and T. truncatus (modified from Ehara 1999).
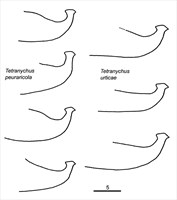
Fig. 16. Comparison of the aedeagi of two cryptic species Tetranychus urticae and T. pueraricola (modified from Ehara & Gotoh 1996).

Fig. 17. Comparison of the aedeagi of two populations of mites identifed as Tetranychus urticae from Australia.
Material examined
non-types
Taxonomy
Subfamily Tetranychinae
Tribe Tetranychini
Common Name
-
Two spotted spider mite
-
Carmine spider mite
-
Common red spider mite
-
Glasshouse red spider mite
-
Hop red spider mite
-
Linden mite
-
Red spider mite
-
Red spider
-
Tropical red spider mite
Distribution
World-wide distribution - see Bolland et al. (1998)
type country = *Germany
Taxonomy Changes
There are many synonymies listed in Bolland et al. (1998) for this species.
The two most recent are:
Acarus haematodes Boisduval 1867
Tetranychus telarius haematodes (Boisduval) Murray 1877, synonymy Smith & Baker 1968
Acarus cinnabarinus Boisduval 1867
Tetranychus cinnabarinus (Boisduval) Boudreaux 1956, synonymy Dupont 1979
Diagnosis
Female
- empodia I-IV with dorsal spur absent above proximoventral hairs (Fig. 1)
- tarsus I with the sockets of four tactile setae proximal to, and one solenidion overlapping, the socket of the proximal duplex seta (Figs 2, 3)
- pregenital striae ranging from longitudinal entire (Fig. 4), to longitudinal with weak or broken striae medially (Fig. 5), to longitudinal with a broad anterior band of broken striae (Figs 6-8)
- peritreme hook moderately long (Fig. 9)
- dorsal striae between setae e1-f1 forming the diamond pattern - i.e. striae between setae e1-e1 longitudinal or often irregular, between e1-f1 transverse, between f1-f1 longitudinal (Figs 10, 11)
- dorsal striae with lobes and ventral striae without lobes
- greenish yellow in cool temperate climate with overwintering orange-yellow form (Gutierrez & Schicha 1983); carmine in warm temperate and subtropical zones (present year round); Davis (1968) lists colour as brownish red, with dark lateral spots to green or yellowish with one large trifid food spot on either side of body (= classic two-spotted mite) and he noted the presence of an orange-reddish overwintering form in Stanthorpe, amongst actively feeding two-spotted females
Male
- empodia I-IV with obvious dorsal spur above proximoventral hairs (Figs 12, 13)
- empodia I with proximoventral hairs fused to form a ventral claw (Fig. 12)
- aedeagus dorsally directed, with narrow neck and distinct knob marginally wider than neck; anterior and posterior projections both short triangular; dorsal margin of knob convex; dorsal margin of shaft at 10° angle to ventral margin, then bending abruptly dorsad at right angle to form knob (Figs 14-17)
- greenish yellow when alive
Hosts
>870 recorded species of host plant: listed in Bolland et al. (1998)
type hosts = *Glycine max (Fabaceae), *Urtica sp. (Urticaceae)
Similar Taxa
Tetranychus parakanzawai Ehara 1999
Tetranychus pueraricola Ehara & Gotoh 1996
Tetranychus truncatus Ehara 1956
Ehara (1999) and Ehara & Gotoh (1996) separate these species based on the length of the knob of the aedeagus (in addition to molecular differences (Gotoh et al. 1998) (Figs 11, 12):
urticae 2.5-2.6 μm; pueraricola 2.1 μm; truncatus 1.5 μm; parakanzawai = 3.3 μm
Biology
Tetranychus urticae is the most damaging spider mite of all. Heavy infestations can destroy crops and kill trees. Control is usually achieved through careful application of pesticide and the use of biological control agents, usually the Chilean predatory mite, Phytoseilus persimilis (Phytoseiidae). Outbreaks of T. urticae are usually associated with the overuse of pesticides.
Adult females are green in areas with cool temperate climate and have overwintering stages that are orange-yellow (Gutierrez & Schicha 1983). In warm temperate and subtropical zones, adult females are carmine and are present year round. Adults overwinter on the ground or in sheltered areas such as bark.
References
Auger, P., Migeon, A., Ueckermann, E.A., Tiedt, L. and Navajas, M. (2013) Evidence for synonymy between Tetranychus urticae and Tetranychus cinnabarinus (Acari, Prostigmata, Tetranychidae): review and new data. Acarologia 53(4): 383-415
Bolland H.R., Gutierrez J., and Flechtmann C.H.W. (1998) World Catalogue of the Spider Mite Family (Acari: Tetranychidae). Brill Academic Publishers, Leiden, 392 pp.
Brandenburg, R.L. and Kennedy, G.G. (1981) Differences in dorsal integumentary lobe densities between Tetranychus urticae Koch and Tetranychus cinnabarinus (Boisduval) (Acarina: Tetranychidae) from north-eastern North Carolina. International Journal of Acarology 7: 231-234
Dupont, L.M. (1979) On gene flow between Tetranychus urticae Koch, 1836 and Tetranychus cinnabarinus (Boisduval) Boudreaux 1956 (Acari: Tetranychidae): synonymy between the two species. Entomol. Exp. Appl. 25: 297-303
Ehara, S. (1956) Tetranychoid mites of mulberry in Japan. Journal of the Faculty of Science, Hokkaido University, Series VI Zoology 12: 499-510
Ehara, S. (1999) Revision of the spider mite family Tetranychidae of Japan (Acari, Prostigmata). Species Diversity 4: 63-141
Ehara, S. and Gotoh, T. (1996) Two new species of spider mites occurring in Japan (Acari, Tetranychidae). Journal of the Acarological Society of Japan 5(1): 17-25
Gotoh, T., Gutierrez, J. and Navajas, M. (1998) Molecular comparison of the sibling species Tetranychus pueraricola Ehara & Gotoh and T. urticae Koch (Acari: Tetranychidae). Entomological Science 1(1): 55-57
*Koch, C.L. (1936) Deutsche Crustacea, Myriapoda, Arachnida, Fasc. I.
Migeon, A. and Dorkeld, F. (2006-2017) Spider Mites Web: a comprehensive database for the Tetranychidae. http://www.montpellier.inra.fr/CBGP/spmweb
Smith, F.F. and Baker, E.W. (1968) Names of the two-spotted spider mite and the carmine spider mite to be redesignated. Co-op. Econ. Insect. Re., U.S. Dept. Agric. 18: 1080
Zhang, Z.Q. and Jacobson, R.J. (2000) Using adult female morophological characters for differentiating Tetranychus urticae complex (Acari: Tetranychidae) from green-house tomato crops in U.K. Systematic and Applied Acarology 5: 69-76
Notes
Tetranychus cinnabarinus and T. telarius are here considered synonyms of T. urticae. Tetranychus cinnabarinus is considered to be the carmine form of T. urticae (Auger et al. 2013). The splitting of the two taxa T. urticae and T. cinnabarinus still occurs in the literature, and their taxonomic status can be somewhat controversial. In some locations the two taxa occur together and are apparenlty distinguishable (Zhang & Jacobson 2000); however a wide degree of reproductive compatability exists between.
Lobes on the striae have been used as a means for separating the two taxa (Brandenburg & Kennedy 1981); however, cuticular lobes are highly variable in their presence/absence and shape.
Copyright © 2018. All rights reserved.