Background
Skipper butterflies are a large, ubiquitous family with approximately 4,200 species globally and 124 named species in Australia. Greatest diversity is in the tropics and they are absent from New Zealand. Most skipper butterflies are dully coloured and small and inconspicuous; the common name relates to their quick, darting flight. In general the immature stages and biology of this group are poorly understood. Caterpillars usually form shelters from the leaves of food plants tied with silk, although the giant skippers are borers. Most skippers by far do not come into conflict with humans and cause economic problems.
Subfamilies
The family currently consists of the following seven subfamilies (Toussaint et al. 2018):
- Coeliadinae
- Euschemoninae
- Eudaminae
- Pyrginae
- Heteropterinae
- Hesperiinae
- Trapezitinae
Short Description
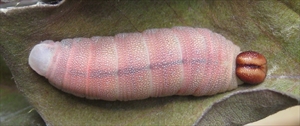
Mature hesperiid caterpillars are ‘grublike’, elongate, cylindrical, tapering at both ends and have numerous, short secondary setae (Fig. 1). Caterpillars have a distinct neck caused by a constriction posterior to the head (Fig. 1, Fig. 2). The head is prominent, except in the giant skippers or Megathymini (Hesperiinae), in which it is always smaller than the prothorax. They are normally greenish, whitish or yellowish. The caterpillars lack an osmeterium. The body segments are divided into annulets (Fig. 1, Fig. 2). Crochets are arranged in a circle. An anal comb is usually present, but not in the Megathymini (Hesperiinae) (giant skippers) (Braby 2000).
Diagnosis
Adapted from Stehr et al. (1987), Ackery et al. 1999.
Diagnostic features are as follows:
- constriction posterior to the head capsule, giving the appearance of a ‘neck’ (Fig. 1, Fig. 2) (except in the giant skippers or Megathymini (Hesperiinae)). This character is unique in the Lepidoptera (Ackery et al. 1999)
- head prominent and often wider than the prothorax (T1), (but never in the giant skippers or Megathymini (Hesperiinae), in which it is narrower than the prothorax) (Fig. 1)
- numerous, short secondary setae on the head and body (Fig. 2)
- often, the distal ends of the setae are flattened
- crochets arranged in a circle
- anal comb sometimes present
Detailed Description
Adapted from Stehr et al. (1987).
General description (see Fig. 1, Fig. 2): The body tends to taper at both ends. There is a constriction posterior to the head capsule, giving the appearance of a ‘neck’ (Fig. 1, Fig. 2) (except in the giant skippers or Megathymini (Hesperiinae)) and the head is prominent and often larger than T1. The integument is variously rugose or wrinkled, and has numerous, short secondary setae (Fig. 2). The position of the primary setae is indicated by rounded or irregularly shaped pinacula. The body is often cryptically coloured and patterned green, brown or white, often with contrasting longitudinal stripes.
Head: The head is hypognathous with the frontoclypeus as long as or longer than wide. It has straight margins extending about one-half the way to the epicranial notch. The ecdysial lines converge about one-fourth to one-half the length of the frontoclypeus above its apex. There are six stemmata with numbers 1-4 evenly spaced in a semicircle and number 5 separated from 1-4 approximately near the antennal base. The location of stemma 6 tends to be variable and always caudad of stemma 4 but may be located dorsad of 4. Stemma 6 may also be further than, equidistant from, or closer to 5 than 4. The primary setae tend to be obscured by the abundant secondary setae making the head appear 'fuzzy' (Fig. 2). The colour of the head is variable but is usually dark and often marked with vertical stripes or with contrasting dots. The integument is often pitted and rugose.
Thorax: T1 is much narrower right behind the head, forming the characteristic ‘neck’. The prothoracic shield is usually well-defined and divided mid-dorsally. It has numerous long setae that may be concentrated towards the anterior margin. Interestingly, clear spots in the shield may indicate the former position of primary setae. The T1 spiracle, sometimes located on the shield, is elliptical and obviously larger than the abdominal spiracles. One or two rounded or irregular plates, dorsal to the leg, possibly indicate the once location of the now absent SV1 and SV2. There are other plates near the spiracle which also may indicate the former presence of other primary setae. Again, some plates, present on T2 and T3 particularly in the subdorsal position may indicate the former position of primary setae. There are numerous long setae on the thoracic legs. The T1-T3 segments are distinctly divided into annulets.
Abdomen: The spiracles on A1-A8 are round or elliptical. The A8 spiracle is larger than and dorsad of the rest of the spiracles. As in the thorax there are rounded or irregular dark rimmed plates on all segments that may indicate the former location of primary setae. Again secondary setae are abundant. The crochets on A3-A6 are bi- or triordinal, and arranged in complete, or slightly incomplete, circles. The crochets on A10 are also bi or triordinal and arranged circularly except for a posterior gap. The dorsal surface of A10 has abundant and long secondary setae. An anal comb is present, except in the Megathymini (Hesperiinae) (giant skippers). Like the thoracic segments, all abdominal segments are divided into annulets. Glands, which may be inconspicuous, are usually present on the venters of A7 and A8.
Species of Biosecurity Concern
THE FOLLOWING SPECIES IS OF BIOSECURITY CONCERN TO NORTHERN AUSTRALIA
Erionota thrax (banana skipper, banana leaf roller)
Four subspecies are recognised:
- Erionota thrax thrax (Indonesia)
- Erionota thrax mindana (southern and central Philippines)
- Erionota thrax hasdrubal (northern Moluccas)
- Erionota thrax alexandra (Luzon, northern Philippines)
Diagnosis
Superficially, the adults of Erionota thrax can be confused with those of the rounded palm-redeye (E. torus), with which it is partially sympatric, and shares a similar biology. Closely related other adults of these species can also be confused, again superficially, with the uncommon E. acroleuca, which has a similar indigenous distribution to E. thrax and E. torus in Asia (see below). There is also another very similar species (on adults) of the genus in the Philippines, E. surprisa (CABI 2019, Cock 2015). At this stage there is no reliable way to separate the larvae of the more important species (from a biosecurity viewpoint), E. thrax, E. torus and E. acroleuca (Cock 2015).
However the larvae of several of these three species can be separated, at least, on food plant (Cock 2015):
- E. thrax and E. torus feed only on Musa spp. (bananas and plantains), and a few other similar species but not palms
- E. acroleuca feeds only on palms (Fig. 1).
All three of these species are considered to be agricultural pests.
Description
Mature caterpillars are cylindrical, with long, pale setae. They are pale green, but often appear white because of an extensive coating of a white, waxy, flocculent powder, which increases with age (Fig. 1, Fig. 3). The head is dark brown to black (Horsfield & Moore 1857), large, wider than the prothorax, and attached by a distinct neck to the body, as is typical of the Hesperiidae (Fig. 1). It is indented at the vertex and widest near it base, appearing heart-shaped from the front. Head setae are similar to body setae. The first thoracic segment, spiracles and prolegs are all a similar colour to the body (Fig. 1). Mature larvae can grow to lengths of 60 mm (Cock 2015, CABI 2019).
Biology and Feeding Damage
Caterpillars form a shelter by cutting an oblique incision in the leaf and forming a tube with this portion of the leaf and fastening it with silk (Fig. 3, Fig. 4). The caterpillars feed inside the rolled leaf, in which black frass accumulates (Fig. 3) (Cock 2015). Rolls can extend up to 150 mm long (CABI 2019).
Leaf damage (Fig. 4), which can lead to defoliation (CABI 2019).
Current Distribution
The indigenous distribution of this species is in Asia, from India through south-eastern Asia to Papua New Guinea, up to southern China.
Pacific Ocean
- Solomon Islands
- Hawaii
Caterpillar Host Plants
- bananas and plantains (Musa, including Manila hemp (M. textilis)
- travellers tree, travellers palm (Ravenala madagascariensis) (Cock 2015)
For more detailed information see: https://www.cabi.org/isc/datasheet/21833.
References
Ackery, P. R., de Jong, R. & Vane-Wright, R. I. (1999). The Butterflies: Hedyloidea, Hesperioidea and Papilionoidea. In: Kristensen, N.P. (Ed.), Handbook of Zoology. 4(35.1). De Gruyter, Berlin, New York.
Braby, M. F. (2000). The Butterflies of Australia: Their Identification, Biology and Distribution. CSIRO Publishing, Victoria, Australia.
Centre for Agriculture and Bioscience International (CABI) (2019). Invasive Species Compendium. Erionota thrax (banana skipper) https://www.cabi.org/isc/datasheet/21833. Accessed July 2019.
Cock, M. J. W. (2015). A critical review of the literature on the pest Erionota spp. (Lepidoptera, Hesperiidae): taxonomy, distribution, food plants, early stages, natural enemies and biological control. CAB International. http://www.cabi.org/cabreviews
Horsfield T, Moore F. (1857). A Catalogue of the Lepidopterous Insects in the Museum of the Hon. East-India Company. Volume I. W.M.H. Allen & Co, London; 1857, v + 278 pp. + IV pp. appendix + 11 pp. index + IX plates larvae + Via plates adults.
Stehr, F.W., Martinat, P.J., Davis, D.R., Wagner, D.L., Heppner, J.B., Brown, M.E., Toliver, M.E., Miller, J.Y., Downey, J.C., Harvey, D.J., McFarland, N., Neunzig, H.H., Godfrey, G.L., Habeck, D.H., Appleby, J.E., Jeffords, M., Donahue, J.P., Brown, J.W. & Frack, D.C. (1987). Order Lepidoptera, pp 288–596. In Stehr, F. W. (Ed.), Immature Insects. Kendall/Hunt, Dubuque.
Toussaint, Emmanuel F. A., Breinholt, E. W., Earl, C., Warren, A. D., Brower, A. V. Z., Yago, M., Dexter, K. M., Espeland, M., Pierce, N. E., Lohman, D. J. & Kawahara, A. Y. (2018). Anchored phylogenomics illuminates the skipper butterfly tree of life. BMC Evolutionary Biology. 18:101 https://doi.org/10.1186/s12862-018-1216-z