Background
The Pyralidae is a large ubiquitous family of 6,000 species. This is a very diverse group of moths with many pest species including the following: pests of beehives; stem-borers; pests of stored food products; defoliators; fruit-borers; seed-eaters; and leaf-webbers. Pyralid moths are small to medium-sized. Caterpillars are almost exclusively concealed feeders. The Pyralidae and Crambidae constitute the superfamily the Pyraloidea. There are 427 named species of Pyralidae in Australia.
Subfamilies
The Pyralidae currently consists of the following five subfamilies:
- Chrysauginae
- Galleriinae
- Pyralinae
- Epipaschiinae
- Phycitinae
Short Description
Adapted from Stehr et al. (1987) and Solis (1999).
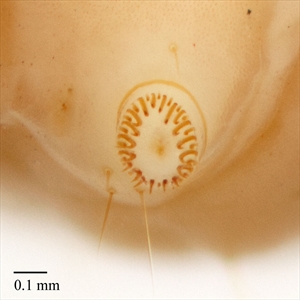
Pyralid caterpillars are generally cylindrical and tapered anteriorly and posteriorly. They have the usual number of prolegs, which can sometimes be reduced but still retain crochets (Fig. 4). Crochets are usually arranged in a circle or modified circle and in the mature larvae are multiordinal (Fig. 2). Well-developed primary setae are present, often from distinct pinacula (Fig. 4). There are two lateral setae on the prespiracular plate of T1 (Fig. 1).
Diagnosis
Diagnosis of the Pyraloidea – Pyralidae and Crambidae
Adapted from Stehr et al. (1987) and Solis (1999).
Almost all pyraloid larvae are concealed feeders e.g. leaf rollers, leaf webbers, leaf miners, borers, root feeders, and seed and fruit feeders, within bird nests, or under silk coverings.
Pyraloid larvae can be distinguished from other Lepidoptera on the following combination of characters:
- Two L setae on the prespiracular plate of T1 (Fig. 1). Pyraloids (pyralids and crambids), noctuoids (consisting of the families: Oenosandridae, Notodontidae, Euteliidae, Erebidae, Nolidae and Noctuidae), and other macrolepidopteran groups have two L setae on the prespiracular plate of TI, whereas many microlepidopteran groups have three L setae in the same location.
- Crochets in a complete circle or penellipse (Fig. 2). Pyraloids can be distinguished from noctuoids on this character because noctuoids usually have the crochets arranged in a mesoseries, whereas crochets, in pyraloids, are in a complete circle or penellipse.
- Three subventral (SV) setae on abdominal segments A3 to A6. Larvae of the Carposinidae also have two L setae on the prespiracular plate of T1 (Fig. 1) and crochets arranged in a circle (Fig. 2). They can be distinguished from pyraloids because they usually have four or more subventral setae, but this may vary from segment to segment.
Pyralidae
Pyralid larvae are concealed feeders with a great diversity of feeding strategies, shelter building and hosts: e.g. stem borers; consumers of stored food products; defoliators; fruit borers; and seed eaters.
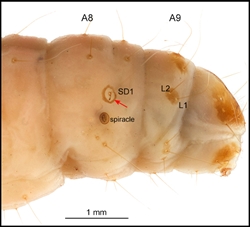
- Sclerotised ring around SD1 on A8 present (Fig. 3). This character distinguishes the Pyralidae from the Crambidae, in which it is absent, although it is missing from the Phycitinae. Note that the ring is sometimes difficult to detect and is sometimes a shiny, white, unsclerotised, translucent ring (e.g. Galleria mellonella (Pyralidae: Galleriinae), greater wax moth (Solis 2007).)
- Three, or occasionally two*, L setae on A9 (Fig. 3). This character distinguishes the Pyralidae from the Crambidae, in which there are only one or two L seta(e) on A9. (*For example, there are only two A9 L setae in both phycitine pyralid species Etiella zinckenella (pulse pod borer moth), which has been introduced into Australia, and carob moth Ectomyelois ceratoniae (Fig. 3).)
Pyralidae Subfamilies
For a key to Pyralidae subfamilies, see Solis (2006), https://www.ars.usda.gov/ARSUserFiles/12754100/PyraloideaKey.pdf.
Diagnosis of Subfamilies
Adapted from Solis (2006).
The presence or absence of sclerotised rings around the bases of specific body setae are important in differentiating subfamilies.
- Chrysauginae
- Sclerotised ring around seta SD1 of the T3 (thoracic segment 3)
- Sclerotised ring around seta SD1 of the T3 (thoracic segment 3)
- Galleriinae
- Sclerotised ring around seta SD1 of A1
- Pyralinae
- No sclerotised ring around the base of seta SD1 on T2 (thoracic segment 2), T3 (thoracic segment 3) or A1.
- Epipaschiinae
- No sclerotised ring around the base of seta SD1 on T2 (thoracic segment 2), T3 (thoracic segment 3) or A1.
- Phycitinae
- Sclerotised ring around seta SD1 on T2 (thoracic segment 2) (most taxa, but some exceptions, notably Etiella).
Detailed Description
Adapted from Stehr et al. (1987) and Solis (1999).
Body (Fig. 4): Cylindrical, tapered anteriorly and posteriorly with usually well-developed prolegs on the A3-A6 and A10. The epidermis of the body is smooth to slightly granular and usually more or less unicolourous, with the venter frequently paler. Tonofibrillary platelets are associated with the epidermis and may be a distinctive pigmented feature on the head, prothoracic and anal shields of many pyraloids. These structures also sometimes occur in other parts of the body as in some Dioryctria (Phycitinae) and Hypsopygia (Pyralinae). The usual complement of primary setae is present in most species but reduced in a few. Most have a sclerotised pinaculum ring with an unpigmented or unsclerotised centre at the base of some setae (Fig. 3). The spiracles are usually elliptical, with those on T1 (Fig. 1) and A8 (Fig. 3) frequently larger.
Head: More or less evenly rounded, and semiprognathous in most species (prognathous and flattened in some Phycitinae (Fig. 1, Fig. 4).) There are six stemmata, but occasionally fewer (Galleriinae). The mandibles are usually simple. The maxillae are usually simple with sensilla trichodea.
Thorax: Usually has a distinct prothoracic shield (Fig. 1), characteristically with six setae on each side. Almost all species have two L setae on the prespiracular plate of T1 (Fig. 1). However, in some, such as Cacotherapia (Galleriinae), the L setae are on the lower margin of the prothoracic shield and are not included in the prespiracular plate. D1 and D2 are present and usually adjacent to each other on T2 and also on T3 (Fig. 1). However, D1 and D2 are somewhat remote in the Galleriinae. SD1 and SD2 are present and adjacent to each other on T2 and also on T3 (Fig. 1). There is a sclerotised and usually darkly pigmented pinaculum ring at the base of SD1 on T2 in most Phycitinae (Fig. 1) or D2 on T3 in most Chrysauginae. The L group is usually trisetose on T2 and T3 (Fig. 1). The SV group is bisetose on T1, and unisetose or bisetose on T2 and T3.
Abdomen: D1 and D2 are usually on all segments, with D1 more dorsad and anteriad than D2 (Fig. 1), except on A9 where D1 is usually ventrad of D2. SD1 is present on all segments. The Galleriinae sometimes have a pinaculum ring around the base of SD1 on A1. On A8, SD1 usually has a sclerotised and usually darkly pigmented pinaculum ring (Fig. 1). SD2 is on all segments except A9 and A10, but is very small. On A1-8, L1 and L2 are usually approximate and more or less ventrad of the spiracle (Fig. 1). L2 setae is usually present on A9 (Fig. 3). On A1-8, L3 is below L1 and L2. SV is bisetose or trisetose on A1-6. SV is unisetose to trisetose on A7-9. V1 is present on all segments.
Crochets are uni- to triordinal on the prolegs on segments 3-6 (Fig. 2). The crochets are most often arranged in the form of circles or penellipses (Fig. 2); in the Chrysauginae they can be incomplete ellipses that are open laterally and mesally. A10 usually has a relatively indistinct suranal plate, usually bearing eight setae. The crochets on the proleg on A10 are usually bi- to triordinal. Typically, nine setae are on and near the anal proleg.
Species of Biosecurity Concern
THE FOLLOWING SPECIES ARE OF BIOSECURITY CONCERN TO NORTHERN AUSTRALIA
Orthaga euadrusalis (mango leaf webber) (Epipaschiinae)
The only known host plant of this species is mango (Mangifera indica). The fully grown caterpillar is brownish blue with whitish striation dorsally. It lacks sclerotised rings around the bases of setae SD1 on T2 (thoracic segment 2), T3 (thoracic segment 3) and A1.
For images see: http://www.cish.res.in/Technologies/Advisory_%20leaf%20webber%20eng%20_2012.pdf
Biology and Feeding Damage
Immature larvae are gregarious, and feed by scraping the top layer of the leaf surface. As larvae mature they begin to feed singly and consume the entire leaf, leaving only the midrib. The caterpillars web the leaves and terminal shoots together to form clusters, which may contain several larvae. The caterpillars feed on leaves, growing points and flowers (CABI 2019).
Feeding Damage
The clusters of leaves die and become dry and brown (Fig. 5).
Current Distribution
- Borneo
- India
Caterpillar Host Plants
• mango (Mangifera indica)
***
Citripestis sagittiferella (citrus fruit borer) (Phycitinae)
No other pyralid caterpillar is known to infest citrus fruits in Asia and Australasia (Anderson 2012).
Description
Caterpillars of this species are orange to dark reddish-brown, with a dark-brown head capsule. Length about 20 mm.
Biology and Feeding Damage
Immature larvae are gregarious.
Caterpillars penetrate the pulp and pith of fruit of citrus trees, causing rotting and premature dropping of infested fruit. Holes in the surface of the fruit are caused by burrowing and used for ejecting frass. Feeding damage can also result in holes in, and cavities under the fruit surface. The larvae are very active when disturbed and jump and twist as an escape mechanism (Anderson 2012).
For more information see: http://www.planthealthaustralia.com.au/wp-content/uploads/2015/01/Citrus-fruit-borer-FS.pdf
Current Distribution
Southeast Asia:
- Indonesia
- Malaysia
- Singapore
- Thailand
Caterpillar Host Plants
- Citrus
References
Anderson, A. (2012) Citrus Fruit Borer (Citripestis sagittiferella) Updated on 2/24/2012 7:21:05 PM Available online: PaDIL - http://www.padil.gov.au.
Centre for Agriculture and Bioscience International (CABI) (2019). Plantwise Knowledge Bank. Orthaga euadrusalis. https://www.plantwise.org/knowledgebank/datasheet/37932. Accessed June 2019.
Gilligan, T. M. & S. C. Passoa. 2014. LepIntercept, An identification resource for intercepted Lepidoptera larvae. Identification Technology Program (ITP), USDA-APHIS-PPQ-S&T, Fort Collins, CO. [accessed at www.lepintercept.org].
Solis, M.A. (1999) Key to selected Pyraloidea (Lepidoptera) larvae intercepted at U.S. Ports of entry: Revision of Pyraloidea in “Keys to some frequently intercepted lepidopterous larvae” by D. M. Weisman, 1986. Proceedings of the Entomological Society of Washington, 101(3), 645–686.
Solis, M.A. (2007) Phylogenetic studies and modern classification of the Pyraloidea (Lepidoptera). Revista Colombiana de Entomología 33(1): 1-9
Stehr, F.W., Martinat, P.J., Davis, D.R., Wagner, D.L., Heppner, J.B., Brown, M.E., Toliver, M.E., Miller, J.Y., Downey, J.C., Harvey, D.J., McFarland, N., Neunzig, H.H., Godfrey, G.L., Habeck, D.H., Appleby, J.E., Jeffords, M., Donahue, J.P., Brown, J.W. & Frack, D.C. (1987) Order Lepidoptera, pp 288–596. In Stehr, F. W. (Ed.), Immature Insects. Kendall/Hunt, Dubuque.